Cardiovascular diseases (CVDs) remain the leading cause of death in the world, according to the World Health Organization, with an estimated 17.9 million people dying from CVDs in 2019. Of these deaths, 85% were due to heart attack and stroke.
Due to the gravity of CVDs and the silent risks that they pose on patients; scientists, biologists and tech founders all over the world are constantly seeking and developing new techniques, sensors and procedures to prevent CVD complications in high-risk patients. The global market is filled with innovative health-tech startups trying to offer early detection, better monitoring or lesser-invasive surgeries for patients with heart diseases.
In this article, we would like to shine the light on two health-tech startups - Bloomer Tech and Meacor - founded by two entrepreneurs of Lebanese Descent residing in North America.
Bloomer Tech: Accelerating Women’s Heart Health
 Although heart disease is sometimes thought of as a man’s disease, it is in fact the cause of death of 1 out of 5 women in the United States alone. Desiring to avoid a fate they know intimately by obtaining more inclusive data around how women present with heart diseases differently than their counterpart and help doctors detect early signs of silent or intermittent heart conditions, Aceil Halaby co-founded Bloomer Tech along with Alicia Chong and Monica Abarca.
Although heart disease is sometimes thought of as a man’s disease, it is in fact the cause of death of 1 out of 5 women in the United States alone. Desiring to avoid a fate they know intimately by obtaining more inclusive data around how women present with heart diseases differently than their counterpart and help doctors detect early signs of silent or intermittent heart conditions, Aceil Halaby co-founded Bloomer Tech along with Alicia Chong and Monica Abarca.
After meeting at MIT, they decided to develop proprietary flexible circuits and sensors embedded inside a woman’s bra to continuously monitorECG, pulse rates, respiratory rates, and more across a woman’s lifespan, all without disrupting a woman's lifestyle. The end product - which was featured in Forbes’ 50 Women-led Startups That are Crushing Tech - is a bra that women may wear under a doctor’s supervision to generate data that will support quality health outcomes and targeted disease management. The goal is to accelerate the effort of inclusive data collection to advance how we care for unique lifestyles, genetic backgrounds and health goals using novel digital biomarkers.
“If we need more data to understand how sex, lifestyle and genetic differences impact disease prevention and prognosis then why not use something women wear every day?” said Aceil Halaby, co-founder and COO at Bloomer Tech. The three founders have already completed early feasibility tests at the MIT IMES CRC and are inviting collaborators who want to support this data collection effort. They are backed by prominent physicians and cardiologists at MIT and other medical centers and are preparing to launch IRB studies this year.
In addition to a physiological monitor, the Bloomer Tech solution provides access to a platform that includes a mobile application and an data analytics service that can corroborate electronic patient recorded outcomes (ePRO) such as symptoms and use them to support research findings with study partners.. Bloomer Tech is actively raising a series A round to proliferate services built out during their seed round to support large-scale research efforts and deliver to early customers..
The founders are currently focused on testing and validating their platform to launch their biomarkers engine. Following FDA clearance, they hope to commercialize their product to medical centers and doctors.
Meacor: the roadmap to a less invasive heart disease treatment

Heart failure is the number one cause of death in the world. A major contributing and serious condition are the mitral and tricuspid valve regurgitation that affect more than 2% of the general population. Given the growing population of elderly patients with multiple comorbidities, the risk for conventional surgery is elevated. Only a fraction of these patients undergo open-heart surgery.
Meacor has developed Cryocinch, a novel catheter-based anchoring device that replicates the open-heart standard of care for mitral and tricuspid valve repair without the need for open-heart surgery. Meacor’s device compensates for the motion of the heart valve by cryo-adhesion, leading to a simpler, shorter, and more robust procedure with better outcomes.
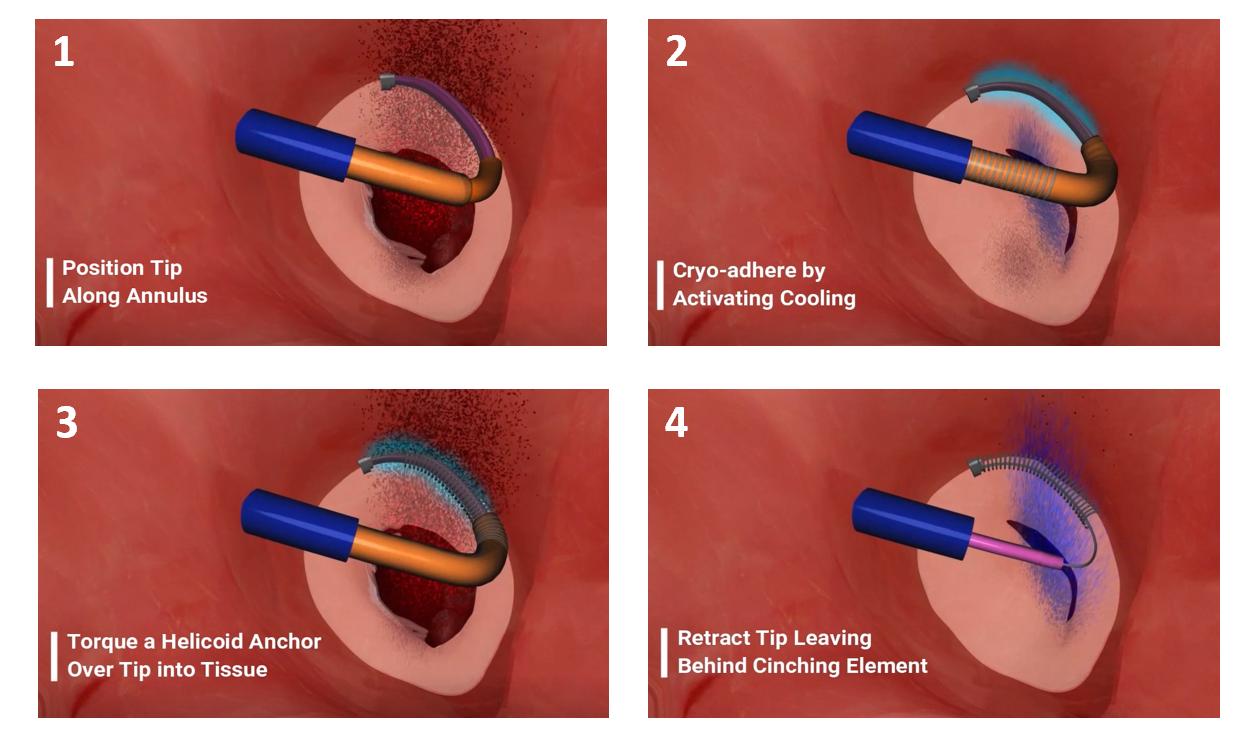
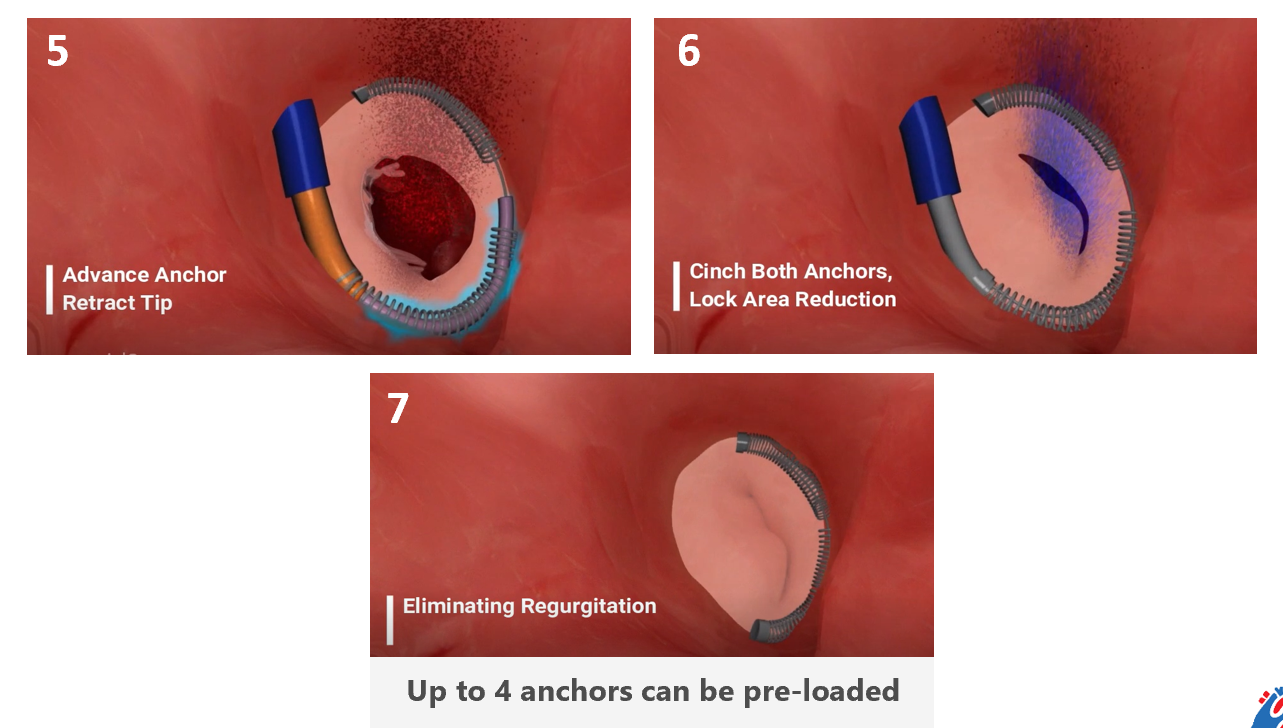
Meacor was co-founded by Toufic Azar - PhD candidate at McGill University in Mechanical Engineering and Dr. Renzo Cecere – Cardiac Surgeon and Professor of surgery at the McGill University Health Center. The team is currently undergoing preclinical trials in live porcine models and is fundraising their series A to transition from the proof-of-concept phase to the development phase. This phase would require 2 to 3 more years to reach design freeze with another 1 to 2 years to reach first-in-human trials. Being a Class III medical device with a lengthy regulatory process, FDA approval is anticipated in 2027-2028.
